Partnership offers quality assurance, more interaction and knowledge exchange
As part of its proposal to offer tools for the constant improvement of laboratory results and in vitro diagnostic products, Controllab offers reagents and equipment suppliers participation in the Collaborating Supplier Program. It is a partnership that provides suppliers and their client laboratories with more interaction and information exchange.
In the program, the supplier makes its products available – reagent, equipment, consumables – for Controllab to perform the repeated analysis sequence for internal control valuation. That is, the reference values for supplier material are now part of Controllab’s internal control programs package, without relying on minimum results formation of laboratories using this supplier’s system.
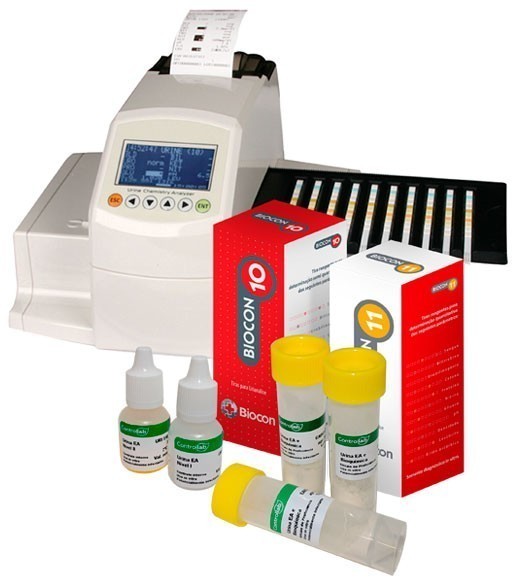
Biocon, which participates in the program, has made its uroanalysis system available for Controllab quality control valuation. At CI ONLINE, Controllab’s Internal Control Program, the laboratory compares its results in real time with other labs that use the Biocon Reader 300 analyzer and routine Biocon 10 and Biocon 11 strips.
Controllab also verifies, with the Collaborating Supplier’s analytical system (kit, reagent, method and/or equipment) the Proficiency Testing (EP) materials, also known as external quality control, a program that allows interlaboratory comparison. By participating in the Controllab EP, the Biocon client laboratory periodically verifies the accuracy of the results for the exams performed with that supplier’s urinalysis analytical system, evaluating its performance against the expected results in the control samples.
External and Internal quality controls allow monitoring processes against different variables that may interfere with the analytical routine: poorly written procedures, inadequate human interface/equipment, batch-to-batch variation, and others. These controls promote reliability in exam results.
“It’s important to note that this partnership is ongoing,” says Jessica Gomes, Controllab Services supervisor. “For each new control material produced by Controllab, whether for use in external control or internal control, analyzes and valuations will be performed in the supplier’s system.” This allows even more integration between the supplier, the client laboratory and Controllab because the program dynamics favor the exchange of information.
In addition, the supplier’s system that participates in the program has quality support because its material is evaluated by Controllab and tested, in some cases, prior to submission of samples, providing opportunities for discussion prior to submission of controls to laboratories. “A laboratory looks for this quality check when choosing a kit, for example. As the collaborating supplier is also a user of Controllab’s internal and external control programs, having this support is another guarantee of his commitment to the quality of the products offered, ” explains Jessica Gomes.
Biocon’s systems as part of the Contributor Supplier Program are continuously evaluated by Controllab, which is an ISO 9001, 17025, 17034, and 17043 recognized Proficiency Test Provider.
Information on how to participate in Quality Control Programs can be obtained directly from Controllab: contato@controllab.com or + 55 21 3891-9900.
